Warning: The oxygenators described on this page and elsewhere on this site are not approved by the FDA for use in humans. Membrane modules should not be used for blood oxygenation in humans under any circumstances.
Long term extracorporeal membrane oxygenation or ECMO (more generally referred to as extracorporeal life support) is a term used to describe prolonged but temporary (1-30 days) support of the heart and lung function using mechanical devices. ECMO can be used for mechanical and respiratory assistance during cardiac and pulmonary failure occurring in newborn infants, older children, or adults. With the outbreak of H1N1 virus (swine flu) in 2009, a significant number of adults and children who developed RDS were successfully treated with ECMO as an alternative to potentially harmful high pressure/ high O2 ventilation.
Blood is drawn from a patient and pumped through an oxygenator where oxygen and CO2 are transferred. The arterialized blood then returns to the patient, either to the venous or arterial circulation depending on the need for cardiac support.
ECMO is almost always partial bypass as opposed to total bypass which is required for CPB operations. With circulation and gas exchange supported mechanically, the heart and lungs are not needed for life support and are allowed to rest. Extracorporeal support continues until the heart or lung function improves. Many days may be required for the heart and lungs to regain adequate function.
Under the auspices of the National Institutes of Health, MedArray has further built upon it's proprietary membrane forming techniques and membrane device designs to improve long term blood oxygenating technology for extracorporeal membrane oxygenation (ECMO) oxygenators. Because MedArray's membrane hollow fibers are made from dense silicone, they are not susceptible to plasma leakage when used for extended periods, unlike microporous membranes which wet-out and leak plasma when used in long term applications. Under NIH SBIR grant, we optimized membrane utilization and flow patterns in three ECMO oxygenator designs suitable for neonatal, pediatric, and adult patients requiring long term extracorporeal support. We used computational fluid dynamics (CFD), gas exchange models, flow visualization, in-vitro, and in-vivo testing to enable the development of greatly improved ECMO oxygenators, suitable for replacing current FDA approved ECMO oxygenators with designs which date back to the early sixties.
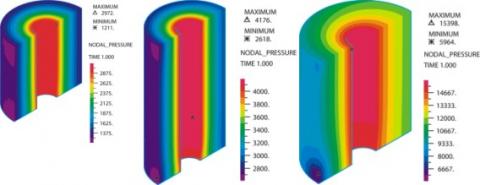
CFD simulations of the blood pressure field in the fiber bundle at rated flow. Note that the isobars are concentric indicating uniform radial flow.
Our results have shown that our three ECMO oxygenators designs can provide the necessary gas exchange and replace six currently commercially available ECMO oxygenators (Medtronic models) with significantly less membrane surface area, blood priming volume, hemodynamic pressure drop, and platelet consumption. The tables below summarize results obtained in Phase II. Our goal is to further improve our designs and to conduct the necessary pre-clinical testing required for an eventual FDA submission for clinical use.
Table 1. Gas exchange performance comparison for MedArray's ECMO oxygenator designs.
| Oxygenator Size |
Membrane Surface Area (m2) |
VO2 per m2 (cc/min/m2) | VCO2 (cc/min) | ||||||
| Medtronic | MedArray | Medtronic | MedArray | Medtronic | MedArray | ||||
|
Neonatal |
0600 | 0.6 |
0.6 |
0600 | 78.3 |
116 |
0600 | 27 |
46 |
| 0800 | 0.8 | 0800 | 87.5 | 0800 | 37 | ||||
| Pediatric | 1500 | 1.5 | 1.1 | 1500 | 76.7 | 92 | 1500 | 65 | 79 |
|
Adult |
I-2500 | 2.5 |
2.8 |
I-2500 | N/A |
129 |
I-2500 | N/A |
221 |
| I-3500 | 3.5 | I-3500 | N/A | I-3500 | N/A | ||||
| I-4500 | 4.5 | I-4500 | 88.9 | I-4500 | 208 | ||||
|
Oxygenator |
Rated Flow (L/min) | Priming Volume (cc) | Pressure Drop at Rated Flow (mmHg) |
||||||
| Medtronic | MedArray | Medtronic | MedArray | Medtronic | MedArray | ||||
|
Neonatal |
0600 | 1.0 |
1.2 |
0600 | 90 |
53 |
0600 | 150 |
24 |
| 0800 | 1.2 | 0800 | 100 | 0800 | 312 | ||||
| Pediatric | 1500 | 1.8 | 1.8 | 1500 | 175 | 98 | 1500 | 185 | 50 |
|
Adult |
I-2500 | 4.5 |
6.5 |
I-2500 | 455 |
202 |
I-2500 | N/A |
211 |
| I-3500 | 5.5 | I-3500 | 575 | I-3500 | N/A | ||||
| I-4500 | 6.5 | I-4500 | 665 | I-4500 | 300 | ||||
Our membrane modules have been used by researchers in experiments requiring blood oxygenation. The following articles have been published.
Dabaghi M, Fusch G, Saraei N, et al. An artificial placenta type microfluidic blood oxygenator with double-sided gas transfer microchannels and its integration as a neonatal lung assist device. Biomicrofluidics. 2018;12(4):044101. Published 2018 Jul 3. doi:10.1063/1.5034791
ABSTRACT Preterm neonates suffering from respiratory distress syndrome require assistive support in the form of mechanical ventilation or extracorporeal membrane oxygenation, which may lead to long-term complications or even death. Here, we describe a high performance artificial placenta type microfluidic oxygenator, termed as a double-sided single oxygenator unit (dsSOU), which combines microwire stainless-steel mesh reinforced gas permeable membranes on both sides of a microchannel network, thereby significantly reducing the diffusional resistance to oxygen uptake as compared to the previous single-sided oxygenator designs. The new oxygenator is designed to be operated in a pumpless manner, perfused solely due to the arterio-venous pressure difference in a neonate and oxygenate blood through exposure directly to ambient atmosphere without any air or oxygen pumping. The best performing dsSOUs showed up to ∼343% improvement in oxygen transfer compared to a single-sided SOU (ssSOU) with the same height. Later, the dsSOUs were optimized and integrated to build a lung assist device (LAD) that could support the oxygenation needs for a 1–2 kg neonate under clinically relevant conditions for the artificial placenta, namely, flow rates ranging from 10 to 60 ml/min and a pressure drop of 10–60 mmHg. The LAD provided an oxygen uptake of 0.78–2.86 ml/min, which corresponded to the increase in oxygen saturation from 57 ± 1% to 93%–100%, under pure oxygen environment. This microfluidic lung assist device combines elegant design with new microfabrication methods to develop a pumpless, microfluidic blood oxygenator that is capable of supporting 30% of the oxygen needs of a pre-term neonate.